Enthalpy - Entropy
Consider a motor piston in the working stroke without ignition, this time additional without the connection to the crankshaft. Because of closed valves
it is thus pushed with from OT to UT from the air, which it has previously compressed. Actually, it could go even further, if the cylinder would only be
indefinitely long.
| This phenomenon has been exploited by
Atkinson for his invention. |
It would come to a standstill, friction would be neglected, if the pressure in the combustion chamber is equal to the ambient pressure. It is only under
these conditions that one can exactly determine the work done, which is called pressure-volume volume work.
Work is defined as work equals force times shift (W = F * s). And since the pressure results as a force through the (pressed piston top) surface, the
work can now be calculated as the area times the shift times the pressure (W = A * s * p). The piston shift times its surface gives a cylinder V. Thus
the pressure-volume work can be determined as the volume change times the pressure (W = ΔV * p).
A nice concept 'volume change work' and also easy to see, since one can clearly imagine the volume gained against the atmospheric pressure.
Considering the combustion chamber as belonging to the system with the displaceable piston surface as one of the system limits, some of the energy
inherent in the system have been used to alter this system boundary.
Actually, the internal energy is called enthalpy H. However, one can not determine it, but only the change of it. And this is achieved by the volume
change. Thus the enthalpy is the sum of internal energy and volume change work (H = U + p * V).
Now and then, imagine that this piston movement would have been infinitely slow. Thus, no pressure increase (isobaric change of state) would have
taken place, because this would have been converted into heat and released to the environment. The process would even be reversible because then
the temperature reduction would have been compensated for by the pressure of the environment.
| Enthalpy is a form of energy, SI unit Joule
(J) |
Processes without pressure change are very frequent. Wherever there is no cover, no enclosed spaces are present, they occur, e.g. during (steel)
melting. They are also available in enclosed spaces. Thus, a large part of the working cycle of the first diesel engine of 1897 continued unchanged
with the final compression pressure of 30-33 bar.
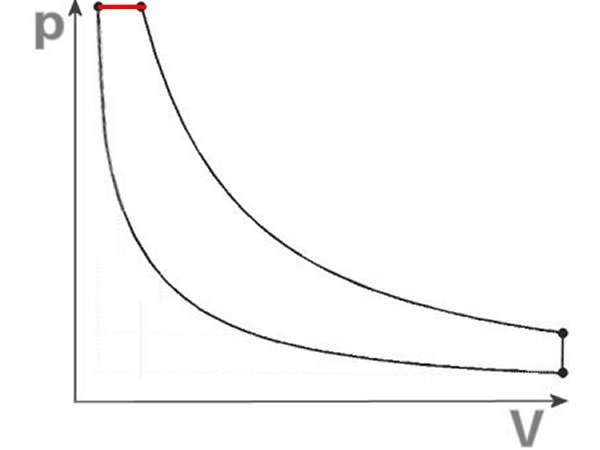
Here are two idealized pV diagrams, calculable curve trajectories, which are supposed to simulate the actual course as well as possible. Above with
four curves, down the Seiliger process with five. Although it is
better adapted, it still contains a piece of isobaric curve (marked red).
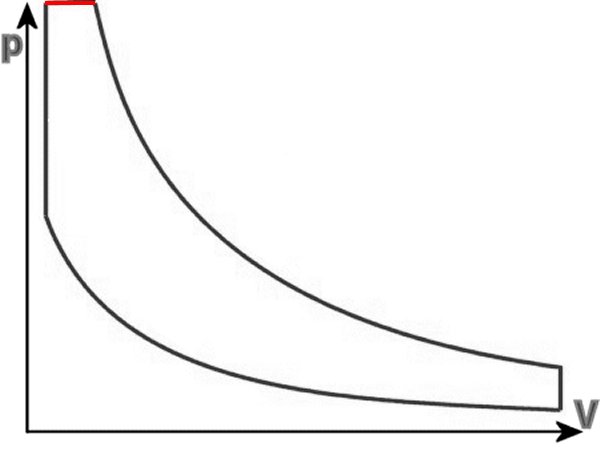
Assumed that the combustion pressure initially develops so slowly that it is absorbed by the volume change through the piston going to UT. Therefore,
the diesel engine is sometimes referred to as a constant pressure motor, whereas the petrol engine because of its electric ignition and other fuel as a
constant volume motor.
Thus, in the very slow process described above, the enthalpy difference can be regarded as the heat transferred to the outside. Perhaps not quite
complete, because, of course, a different distribution (order) is produced by the volume change, even if it is particularly slow.
| Entropy S in enthalpy devided by absolute
(!) temperature (J/K). |
And this is exactly what the entropy explained above, which is explained very vividly in the video. Is more important in the case of (ideal) gases
because their position and order in the space can be a million times different. The precise detection of energy currents during the operation of
thermodynamic systems is of great importance not only for the detection of the efficiency.
However, measures to save fuel or coke are at least 200 years old. Even the invention of the steam engine by James Watt (1736 - 1819) was actually
the improvement of the invention by Thomas Newcomen. Otto's four-stroke engine was the energetic improvement of the Lenoir engine from 1860 and
the diesel engine at least initially replaced the steam engine.
And what does entropy have to do with temperature? The movement of the molecules in gases increases with rising temperature. This reduces the order and increases the entropy. That is what the second principal
of thermodynamics says: Voluntary change of state increases the entropy. 02/17
|