 Exhaust - Emissions 1 Exhaust - Emissions 1
| Single carburettor with extended idle system . . . |
kfz-tech.de/PAg1
It's always assumed that tightened emissions regulations dated back to the early 1970's, but California has been battling smog already since the 1940's. At that time, Europe was still a long way from the peak of mass
motorization. On the contrary, the terrible Second World War still stood against it.
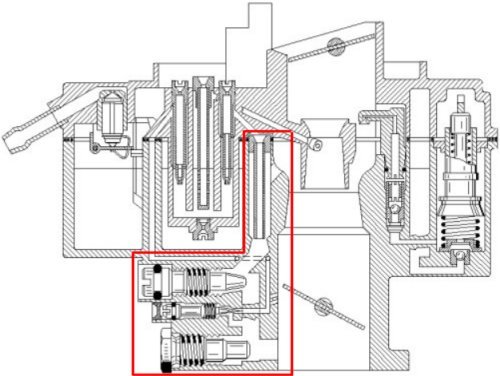
Those who worked in a manufacturer's branch first encountered such regulations on new vehicles around 1970. One was amazed at the quite complex idling systems (picture above) with the corresponding setting
regulations.
That idling could have such an impact on the environment was hard to imagine at the time. In the case of truck diesel engines, it was often still the case that the idling was set correctly when the driver's door seemed to be
relatively calm when it was open. Unimaginable today, just like back then, what all was to come.
As I said, California was 10 years earlier with the legislation and even Japan had been working on the problems a little longer. As an exception, the problem of avoiding carbon monoxide was once approached from the
humor (negro) side by saying that from now on it will be more difficult for suicidal persons to kill themselves in the garage, which is isolated from the outside, with the vehicle in idling.
Which brings us to the first and probably the most dangerous pollutant, carbon monoxide (CO). Carbon is tetravalent and oxygen is bivalent. While searching for the second oxygen atom, CO can attach itself to the red blood
cells and prevent them from absorbing oxygen. The transport of oxygen to the brain is severely impaired.
In addition to the externally perceptible suffocation, there is also an internal one, in which the brain is probably the first to switch off. CO is invisible and odorless. Without a doubt, it is also clear how CO is formed. It must be a
combustion that involves too little air or oxygen. Such a mixture with too much fuel is called a rich mixture (top left).
Hydrocarbons (HC or CH) are also formed when the mixture is too rich. Although they are still harmful to health, they are not as dangerous as carbon monoxide. It is therefore correctly called the carbon monoxide and the
hydrocarbons because a large number of compounds are covered by this generic designation. Tetravalent carbon, mostly chained, together with monovalent hydrogen results in an almost infinite number of possible
combinations.
Exactly the opposite is the case with nitrogen oxides. They occur with a rather lean mixture (top right). Lean mixtures are usually hotter than rich mixtures. This may seem surprising, but it has to do with the higher proportion
of evaporating fuel, which in turn removes an enormous amount of evaporation heat from combustion. In the past, e.g. in racing, a richer mixture was used for cooling at full load.
Nitrogen oxides are therefore formed at temperatures well above 1000°C. Here, the plural 'nitrogen oxides' refers to various compounds, e.g. NO2 and NO3, which are summarized under the
chemical name NOX. Among other things, one ascribes to it the already mentioned formation of smog. Surprisingly, however, the forest had not been harmed as badly as originally thought.
The nitrogen oxides are very important, e.g. for the export of diesel vehicles to the USA or their manufacture and sale there. The Americans applied the same NOX limit values for petrol and diesel engines, while
in Europe diesel was spared a little in this respect for a long time. This is one of the reasons why the diesel engine only very slowly established itself in the passenger car sector there.
However, since Euro 6 this has ended. The transport industry in particular is groaning under the financial burden of new vehicles with this emissions standard. There are discussions about possibly even slightly increased
consumption compared to Euro 5. This may give you an idea of the connection between hot combustion and low fuel consumption. If this is combated internally in the engine, there is a risk of more instead of less
consumption. But of course there have already long been ways of avoiding this as well.
Of course, sulfur oxides depend on the sulfur content in the fuel. Here the group is a bit larger, because in addition to the pure oxides (SOX), there are also the sulfuric and sulphurous acids
(H2SO4 and H2SO3). Of course, the sulfur can't come from anywhere other than the fuel. Its removal there to the last remnant is presented by the refineries as expensive.
Apparently, it is easier to remove from petrol, where the legally prescribed limit values are currently less than half as high. In case of diesel fuel, it poses a problem insofar as it ensures that regeneration is required much
more frequently in the NOX reducing catalytic converter, which of course costs fuel. Sulfur is also suspected of forming acid rain and can cause skin irritation and swelling.
Soot by itself wouldn't even be that bad, since mankind has been grappling with it for thousands of years. Soot is pure carbon, caked together because no oxygen could get to the carbon in the fuel. But unfortunately the soot
also drags other compounds with it that were formed during combustion. And it is precisely then that the soot particles are called 'particles'.
We cannot spare you the designations entirely. This are namely the polycyclic aromatic hydrocarbons. They are strongly suspected of being carcinogenic. At this point, the second task of the oxidation catalytic converter in the
diesel exhaust system, in addition to heating, can also be explained. Since the diesel engine works with excess oxygen, it is obviously necessary to at least partially oxidize these soot appendages and thus render them
harmless.
|