
|
 Chemistry - Periodic Table 1 Chemistry - Periodic Table 1

Finally a subject, in which there is an order or at least a signpost to such an order, the periodic table. It lists all possible atoms, not simply one after the other, but within the two-dimensional space with
special consideration of certain properties. In this book, we also want to take this groundbreaking system as a starting point whenever possible.
It is well known that atoms consist of protons in the nucleus, neutrons, too, except for hydrogen and electrons in a comparatively large shell. Protons are positive, electrons negatively charged, neutrons
none of them. The number of protons and electrons in an atom is always the same. In terms of weight, however, both are very different. With certain considerations we will even consider the weight of an
electron as negligible.
If two atoms have the same number of protons, they are assigned to the same element. If only the number of neutrons deviates from the normal number for this element, one speaks of a 'isotope' of this
element. In the periodic table above 118 elements are listed, line by line from the lowest to the highest number of protons.
For each element there is a panel with a large symbol in the middle, usually the first letter(s) of the Latin name, as with hydrogen the 'H' for hydrogenium. In the upper left corner the number of protons in the
nucleus is indicated, also called 'atomic number'. Since the elements are ordered according to the number of protons, also follow atomic numbers this schema.
And then there is another number on the upper left, in our picture omitted for reasons of clarity, of which at least for this chapter we only want to explain the rounded variant. It includes the number of
particles in the nucleus, i.e. protons and neutrons together. If you now look at the individual series of the periodic table, then from left to right, the number of protons and neutrons increases. atomic number
(number of protons) always by 1. At the end you change to the next row.
And what causes the clearly visible gaps in this system? This is the attempt to arrange the individual elements within the rows in such a way that elements with similar chemical properties are interrelated.
If you would like to count through them, eight different properties are distinguished. In the next line they turn back periodically like the four seasons. Now you know why the system is called 'periodic'.
There are many size comparisons of atomic nuclei and shells. We have one of our own, namely the observation of a football from about 2 km distance, which of course is not possible. But in such a way
approximately you must imagine the relationship. Thus the shell becomes the all-determining part for the size of an atom. But how does such a shell look like at atoms with different order-numbers?
We can still conclude briefly that, because of the compensation of electrical effects, there must be as many electrons in the shell as protons in the nucleus. But if we now start to summarize these
electrons in groups and to assign individual distances or circular paths, additional explanations are necessary. Because already with the past explanations we have for reasons of the transparency partly
simplified.
As an example it may be considered here that further material was discovered in the atomic nucleus, which would have destroyed here however the beautiful simpleness of only three particles, from which
all atoms consist and which differ only in the number of them. If you explain so easily and simply, you only have to make sure that you don't have to relearn when you get to know things better. Because
that's how it is here, in this case you can leave out the special features.
We are now doing the same for the rest of the procedure by using the shell model of Bohr. As already said, it is a model, so it has only limited to do with reality. It explains the part you need it for right now,
maybe more, but you don't know that. So it's probably not the case that electrons race in orbits. Above all, there is no guarantee that you will always be able to know where they are at the moment.
You will already notice that if we continue there now, the connection to the atomic model worked out so far will be completely lost. So let's go into the depths of Bohr's atomic model, probably knowing that
this only accounts for a limited part of the reality, does not really picture.
Starting with hydrogen, we look for the only counterpart to the single proton in the nucleus. It is an electron on the innermost shell, also called 'K-shell'. The so-called shells exist here, because by no
means every electron has its own distance to the nucleus. Rather, the electrons appear in different groups, whereby here we have the same distance to the nucleus for each electron of a group.
You can perhaps imagine that this are about gravitational forces that balance centrifugal ones by the movement of electrons. And these forces are naturally all the more the closer the group is to the core.
Even the number of electrons in a group determines this proximity. But there is one iron basic principle above these assumptions, namely that the energy is also said to be 'quantized'.
This means, if an electron changes the group or the shell e.g. to the inside, then the energy level also changes. It is stepped, so it does not increase evenly. This is the beginning and content of a newer
physics or chemistry, which exists since about 120 years and which was founded by Max Plank, Albert Einstein and others, so there are no possible whereabouts for electrons. outside the shells.
The periodic table is thus constructed according to the Bohr atomic model. Whenever a new shell is needed for additional electrons, a new line is used. And since the innermost shell can only have a
maximum of two electrons, only two elements, hydrogen and helium, are represented in the first line of the periodic table.

And why is the hydrogen on the far left and the helium on the far right? This is because here there are not only seven horizontal periods, but also 18 vertical groups. Don't worry, the 18 can be simplified
considerably. However, we can already state that the attempt has been made to combine elements with similar chemical properties for the respective vertical.

| The noble gases are marked here. |
When do elements have matching chemical properties? This depends on the occupancy of the outer shell. You already know that this is the first shell for hydrogen and helium and one more for each
additional row. Actually, the maximum possible occupancy of the bowls looks like this:
| 1 | K-shell | 2 Electrons |
| 2 | L-Shell | 8 Electrons |
| 3 | M-shell | 18 Electrons |
| 4 | N-shell | 32 Electrons |
| 5 | O-shell | (50) Electrons |
| 6 | P-shell | (72) Electron |
| 7 | Q-shell | (98) Electrons |
And why is the respective number of electrons bracketed in the last three rows? Because these values are never reached by any element. A certain order is kept only up to row 3. In rows 2 and 3 there are
eight elements each. The number of protons must change from element to element, which happens here on the outermost shell. From left to right always increasing by 1:
| Period 2: L- and K-shell |
| Lithium | 2, 1 |
| Beryllium | 2, 2 |
| Boron | 2, 3 |
| Carbon | 2, 4 |
| Nitrogen | 2, 5 |
| Oxygen | 2, 6 |
| Fluor | 2, 7 |
| Neon | 2, 8 |
| Period 3: L-, K- and M-shell |
| Sodium | 2, 8, 1 |
| Magnesium | 2, 8, 2 |
| Alumium | 2, 8, 3 |
| Silicon | 2, 8, 4 |
| Phosphor | 2, 8, 5 |
| Sulphur | 2, 8, 6 |
| Chlor | 2, 8, 7 |
| Argon | 2, 8, 8 |
With row 4 begin the difficulties with this order, because one has only these eight possibilities on the outermost shell. More than eight electrons are not possible there. So what to do if there are now 18
elements in a row. The solution offers a rather idiosyncratic change on the second last shell:
| Period 4: L-, K-, M- and N-shell |
| Potassium | 2, 8, 8, 1 |
| Calcium | 2, 8, 8, 2 |
| Scandium | 2, 8, 9, 2 |
| Titanium | 2, 8, 10, 2 |
| Vanadium | 2, 8, 11, 2 |
| Chrome | 2, 8, 12, 2 |
| Manganese | 2, 8, 13, 2 |
| Ferrous | 2, 8, 14, 2 |
| Cobalt | 2, 8, 15, 2 |
| Nickel | 2, 8, 16, 2 |
| Copper | 2, 8, 17, 2 |
| Zinc | 2, 8, 18, 2 |
| Gallium | 2, 8, 18, 3 |
| Germanium | 2, 8, 18, 4 |
| Arsenic | 2, 8, 18, 5 |
| Selen | 2, 8, 18, 6 |
| Bromine | 2, 8, 18, 7 |
| Krypton | 2, 8, 18, 8 |
Can you see the recent changes? From the third element, in this case Scandium, the last but one shell changes. It begins with 8 electrons in such a way that finally 18 electrons are reached with zinc,
which corresponds to its usual equipment if at least one more shell follows. Only from gallium the outermost shell grows, again up to 8 for the noble gases on the right.
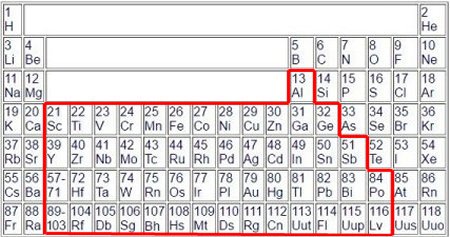
The question remains why the penultimate shell is counted up from the third element in the period. Answer: Because the following ten elements all have very similar properties, they are all metals. So it
remains with their two electrons on the outermost shell. However, although they are still metals, counting is also continued presumably, so that at the end again 8 electrons come out on the outermost
shell.
| Period 5: L-, K-, M-, N- and O- shell |
| Rubidium | 2, 8, 18, 8, 1 |
| Strontium | 2, 8, 18, 8, 2 |
| Yttrium | 2, 8, 18, 9, 1 |
| Zirconium, Nicobium, Molybdenum, Technetium, Ruthenium, Rhodium, Palladium, Silver |
| Cadmium | 2, 8, 18, 18, 2 |
| Indium | 2, 8, 18, 18, 3 |
| Tin, Antimony, Tellurium, Iodine |
| Xenon | 2, 8, 18, 18, 8 |
And since the number of elements per row does not change, the regulation of row 5 can be maintained for the following ones. Here we show only row 6 to point out another problem. Together with Lathan the
Lanthanoids are added, which is taken into account in the table. Similar things happen in the last row with Actinium and the Actinoids. We come back to this when talking about metals.
| period 6: L-, K-, M-, N-, O- and P-shell |
| Cesium | 2, 8, 18, 18, 8, 1 |
| Barium | 2, 8, 18, 18, 8, 2 |
| lanthanum | 2, 8, 18, 18, 9, 2 |
| Cer, Praseodymium, Neodymium, Promethium, Samarium, Europium, Gadolinium, Terbium, Dysprosium, Holmium, Erbium, Thulium, Ytterbium, Lutetium, Hafnium,
Tantalum, Tungsten, Rhenium, Osmium, Iridium, Platinum, Gold |
| Mercury | 2, 8, 18, 32, 18, 2 |
| Thallium | 2, 8, 18, 32, 18, 3 |
| Lead, Bismuth, Polonium, Astat |
| Radon | 2, 8, 18, 32, 18, 8 |
|
|