|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
A B C D E F G H I J K L M N O P Q R S T U V W X Y Z
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Previous page |
| 1 H | 2 He | ||||||||||||||||
| 3 Li | 4 Be | 5 B | 6 C | 7 N | 8 O | 9 F | 10 Ne | ||||||||||
| 11 Na | 12 Mg | 13 Al | 14 Si | 15 P | 16 S | 17 Cl | 18 Ar | ||||||||||
| 19 K | 20 Ca | 21 Sc | 22 Ti | 23 V | 24 Cr | 25 Mn | 26 Fe | 27 Co | 28 Ni | 29 Cu | 30 Zn | 31 Ga | 32 Ge | 33 As | 34 Se | 35 Br | 36 Kr |
| 37 Rb | 38 Sr | 39 Y | 40 Zr | 41 Nb | 42 Mo | 43 Tc | 44 Ru | 45 Rh | 46 Pd | 47 Ag | 48 Cd | 49 In | 50 Sn | 51 Sb | 52 Te | 53 I | 54 Xe |
| 55 Cs | 56 Ba | 57- 71 | 72 Hf | 73 Ta | 74 W | 75 Rn | 76 Os | 77 Ir | 78 Pl | 79 Au | 80 Hg | 81 Tl | 82 Pb | 83 Bi | 84 Po | 85 At | 86 Rn |
| 87 Fr | 88 Ra | 89- 103 | 104 Rf | 105 Db | 106 Sg | Bh 108 | 108 Hs | 109 Mt | 110 Ds | 111 Rg | 112 Cn | 113 Uut | 114 Fl | 115 Uup | 116 Lv | 117 Uus | 118 Uuo |
These are the inert gases. Their name is derived from the fact that they have the property, of not reacting with any other element. On the outside shells of their atoms all the places are occupied by electrons, a special status in the periodic system. We can perceive inert gases then, when a current flows through them and they light up in the most wonderful colours. This occurs because the current-flow causes their molecules to oscillate. Apart from Argon, they are relatively seldom.
Although it could be slotted in well at the end of the periodic system, the last natural occurring element was only discovered at the end of Mendelejew's creative period. This is the radioactive Radon. In the automotive field, apart from being used for lighting purposes, inert gases are also used as shielding gases. In this case, e.g., as far as welding is concerned, they prevent other atmospheric gases from reaching the weld. 03/13

| Helium |
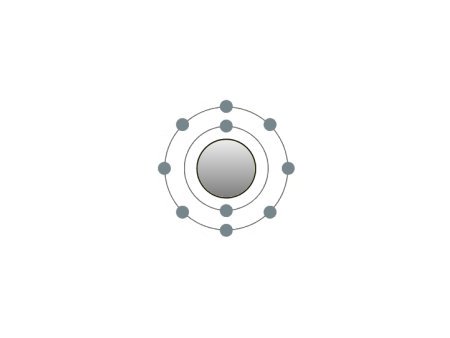
| Neon |
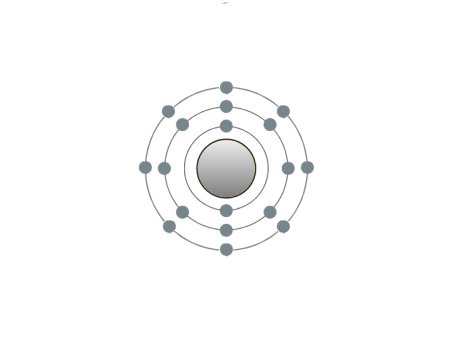
| Argon |
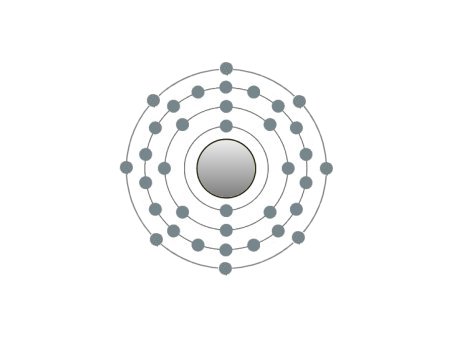
| Krypton |
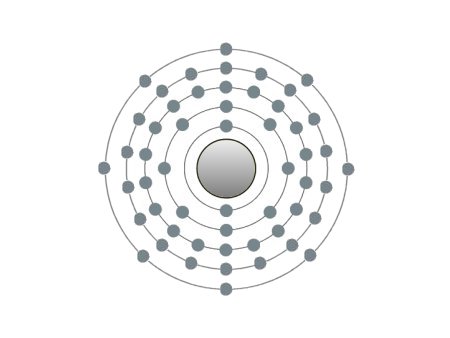
| Xenon |
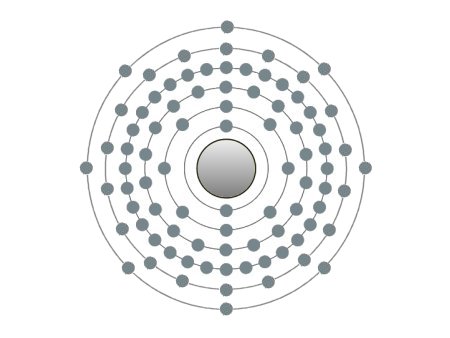
| Radon |
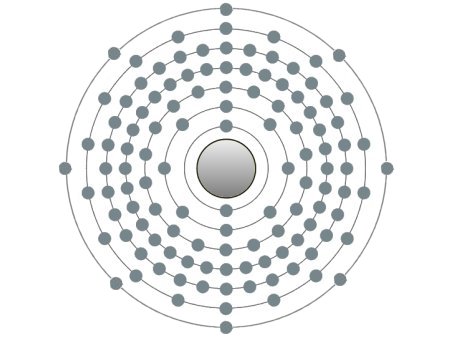
| Ununoctium |
| If the atomic core was the size of a cherry, the entire atom would be as big as the Cologne Cathedral. |
| Next page |
2001-2015 Copyright programs, texts, animations, pictures: H. Huppertz - E-Mail
Translator: Don Leslie - Email: lesdon@t-online.de

