
|
Isentropic Compression
| Much more information about thermodynamics including aggregat states you find here |
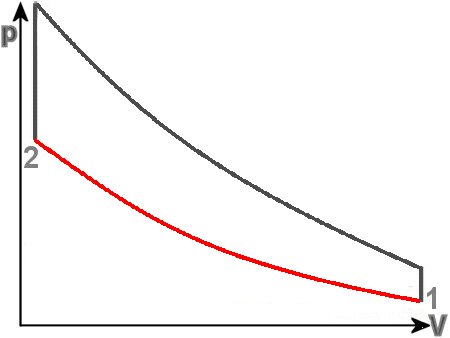
Let us take a look at the compression stroke, indicated by the red in the thermodynamic circle of a gasoline engine. The volume is significantly reduced and the pressure is thereby increased in some way. The
process should be carried out so quickly that there is no heat flow into the environment. The entropy thus remains the same. Under this assumption, the process is also reversible.
We proceed from a single displacement of 500 cm3, as Mini and BMW three- or four-cylinders. On the loader, however, we have to dispense. Nevertheless we begin with moderate 10:1 compression,
which ultimately results in a volume of 50 cm3.
V1 = 500 cm3, V2 = 50 cm3, T1 = 373 K (= 100°C)
T2 = ?
Result: T2 = 937 K (664°C)
The results are obtained by taking the value 1.4 for 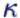 (air) . More about (air) . More about 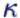 (kappa) can be found further down the page. This results in (kappa) can be found further down the page. This results in 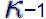 0.4. Now you need an easy to find
(scientific) calculator with the possibility to get xY. 0.4. Now you need an easy to find
(scientific) calculator with the possibility to get xY.
| 9 : 1 | 898 K (625°C) |
| 10 : 1 | 937 K (664°C) |
| 11 : 1 | 973 K (700°C) |
| 12 : 1 | 1.008 K (735°C) |
| 13 : 1 | 1.041 K (768°C) |
| 14 : 1 | 1.072 K (799°C) |
| 15 : 1 | 1.102 K (829°C) |
| 16 : 1 | 1.131 K (858°C) |
| 17 : 1 | 1.158 K (885°C) |
| 18 : 1 | 1.185 K (912°C) |
| 19 : 1 | 1.211 K (938°C) |
And what do you think now? Quite a lot of temperature only for a compression of 10:1. The additionally calculated values give you an idea why a diesel engine with even higher compression can ignite without electrical
spark plugs, if a completely new type of internal combustion engine is being built for the first time, the engine is towed by an electric motor. Only by compression it reaches its operating temperature without burning a
drop of fuel.
Isentropic exponent 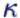 . . . . . . |
Repetition: The specific heat capacity is the energy expenditure in kJ, which is necessary to heat one kg of a substance by one degree. For gases, there are the two possibilities to keep the volume constant or to use a
part of the heat energy supplied to the volume change. The isentropic exponent 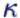 is thus computed by division of cp and cV
the two heat capacities. is thus computed by division of cp and cV
the two heat capacities.
It is logical that more energy is needed in the case of volume change work at cp. If, on the other hand, the volume is constant at cV,
the heat supplied alone benefits the internal energy. cp is always greater than cV, 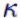 is greater than 1. 02/17 is greater than 1. 02/17
More about 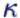 in the video below. in the video below.
|
|
|